|
Back to Blog
Hcn molecular geometry6/17/2023 ClF3 is a polar compound.\) Some examples of polar molecules based on molecular geometry (HCl, NH3 and CH3Cl). This molecule has two lone pairs and three bound pairs, according to the ClF3 Lewis structure. 
What is CLF3 molecular geometry?ĬlF3 has a T-shaped molecular geometry and trigonal bipyramidal electron geometry. As a result, dipole-dipole interactions are observed in hydrogen sulfide. In the case of H2S, the vectorial sum of the bond dipole moments results in a non-zero total dipole moment. It belongs to the cyano group and consists of carbon and a nitrogen atom having a triple bond. The molecule is slightly polar because sulfur is more electronegative than hydrogen. CN is known as cyanide which exists as a pseudohalide anion. The molecule bends due to the existence of two unbonded pairs of electrons. The molecular geometry or shape of the HCN molecule is linear. Because of its electronegative value, nitrogen tries to pull electrons to itself, making it slightly polar. Draw the Lewis structure for the hydrogen cyanide molecule, HCN. The HCN Lewis structure comprises three different atoms: hydrogen, carbon, and nitrogen. According to the VSEPR theory, if the HCN compound has an AX2 generic formula, the molecular geometry and electron geometry will both be linear molecular geometry. Because hydrogen and nitrogen are usually far apart, HCN takes on a linear shape. Hydrogen Cyanide (HCN) is a colorless, flammable, and poisonous liquid. 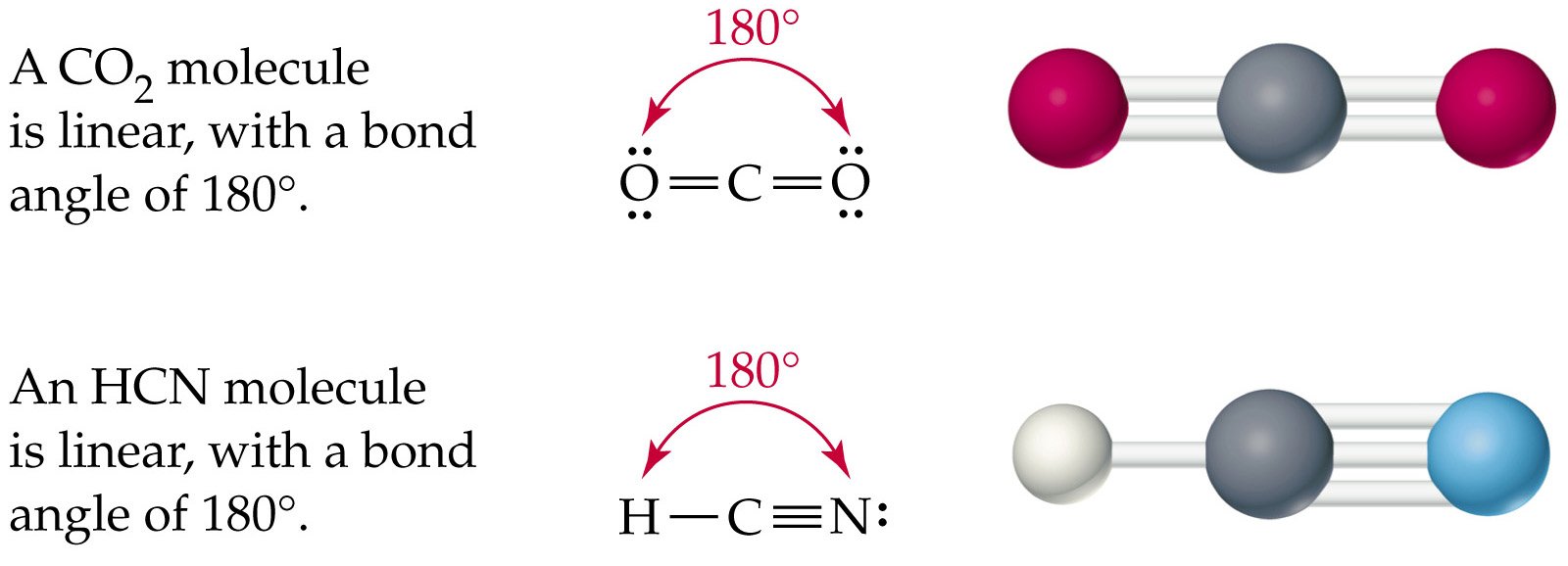
On both sides of the central sulfur atom in the H2S Lewis structure, there are two hydrogen atoms. Hydrogen cyanide has a linear molecular geometry with 180-degree bond angles. What is the dot structure of hydrogen sulfide? Table 1 Molecular geometries along the minimum energy path. Geometry, De value and potential constants of HCN HF 215 in some cases. chemical reactions, as exemplified by the isomerization reaction HNCHCN. In its molecule, both oxygen atoms have the same electronegativity value, both atoms share equal ratios of bonded shared electrons, and the overall O 2 molecule turns out to be nonpolar. The parameters of the molecular potential function determined for. Oxygen is a diatomic nonpolar molecule with a bond angle of 180 degrees. In the O2 Lewis structure, there is a double bond between two oxygen atoms. Determine the electron geometry (eg) and molecular geometry (mg) of BCl3. The nitrogen gains a partial negative charge, while the hydrogen gains a partial positive charge.Īs a result, positive and negative poles are formed across the molecule, and HCN becomes a polar molecule. eg trigonal planar, mg tetrahedral eg linear, mg tetrahedral o eg trigonal planar, mg trigonal planar O eg tetrahedral, mg linear o eg linear, mg linear. This results in unequal charge sharing in the linear-shaped HCN molecule and a non-zero dipole moment. 100 (3 ratings) Transcribed image text: QUESTION 5 Determine the electron geometry (eg) and molecular geometry (mg) of HCN. Click on the image above to be taken to the WebMO. Predict the electron-pair geometry and molecular geometry of the XeF 4 molecule. Whereas carbon and nitrogen form a triple bond to share three electrons. Predicting Electron-pair Geometry and Molecular Geometry: XeF 4 Of all the noble gases, xenon is the most reactive, frequently reacting with elements such as oxygen and fluorine. The difference in electronegativity between atoms is directly proportional to the polarity of the molecule.Ĭarbon is in the center, surrounded by nitrogen and hydrogen atoms.Ĭarbon and hydrogen share electrons to form a covalent bond. 
As can be seen from the HCN Lewis structure, the electronegativity difference between nitrogen (3.04) and hydrogen (2.2) makes it a polar molecule. Hydrogen cyanide is a one-carbon compound consisting of a methine group triple bonded to a nitrogen atom It has a role as a human metabolite, an Escherichia.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
